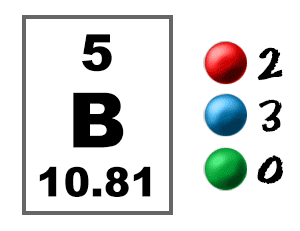
The isotope boron-10 is good at absorbing neutrons. When an organism dies, it stops taking in carbon-14, so the ratio of carbon-14 to carbon-12 in its remains, such as fossilized bones, will decline as carbon-14 decays gradually to nitrogen-14 2 ^2 2 squared. One atomic mass unit is equal to 1.66 x 10 grams. Element Boron (B), Group 13, Atomic Number 5, p-block, Mass 10.81. As animals eat the plants, or eat other animals that ate plants, the concentrations of carbon-14 in their bodies will also match the atmospheric concentration. As plants pull carbon dioxide from the air to make sugars, the relative amount of carbon-14 in their tissues will be equal to the concentration of carbon-14 in the atmosphere. Example 1 The natural abundance for boron isotopes is: 19.9 10 B (10.013 amu) and 80.1 11 B (11.009amu). One atomic mass unit is equal to 1.66 x 10 -24 grams. based on the number of isotopes in the problem. The thermal neutron cross section of boron 10 is 3837 barn, and the thermal neutron cross section of boron 11 is less than 0.1 barn1. The natural abundance of boron 10 is about 19.8, and the natural abundance of boron 11 is about 80.2. The unit of measure for mass is the atomic mass unit (amu). Natural occurring boron has two stable isotopes: boron-10 and boron-11. Therefore this resulting atomic mass is calculated from naturally-occurring isotopes and their abundance. 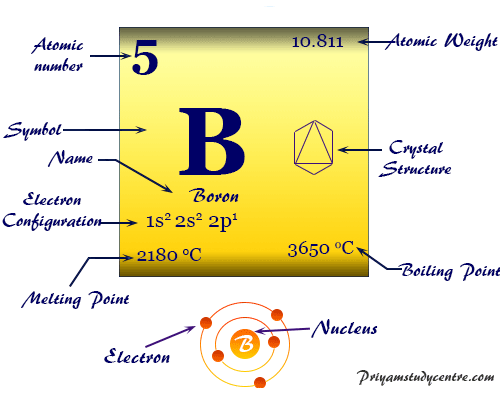
The boron atom has a radius of 90 pm and a Van der. 
These forms of carbon are found in the atmosphere in relatively constant proportions, with carbon-12 as the major form at about 99%, carbon-13 as a minor form at about 1%, and carbon-14 present only in tiny amounts 1 ^1 1 start superscript, 1, end superscript. Note that each element may contain more isotopes. The number of electrons in each of borons shells is 2, 3 and its electron configuration is He 2s2 2p1. For example, carbon is normally present in the atmosphere in the form of gases like carbon dioxide, and it exists in three isotopic forms: carbon-12 and carbon-13, which are stable, and carbon-14, which is radioactive.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
